CHRONO-LOG Model 700 Whole Blood/Optical Lumi-Aggregometer
The CHRONO-LOG® Model 700 Whole Blood/Optical Lumi-Aggregometers are available in a two or four channel configuration. This state-of-the-art Lumi-Aggregometer measures platelet function on patient samples using electrical impedance in whole blood samples or optical density in plasma with the simultaneous measurement ATP Release by the luminescence method. The Model 700 Lumi-Aggregometer also has the capability of performing the Ristocetin Cofactor Assay for diagnosing patients with von Willebrand’s disease. The output can be connected to either a strip chart recorder or to a Computer. AGGRO/LINK®8 and vW Cofactor Software packages are provided for the computer interface option.

With a proven track record of providing accurate and reliable results, the Chrono-log Whole Blood/Optical Lumi-Aggregometer is both a reliable diagnostic tool and a comprehensive screening tool. Direct measurement of ATP secretion during platelet aggregation provides unequivocal evidence of normal dense granule release. The simultaneous measurement of Aggregation and dense granule release provides a better insight into the mechanism of platelet response.
The Model 700 Whole Blood/Optical Lumi-Aggregometer available Housed in a smaller chassis, the Model 700 Aggregometer is available in configurations of up to Four (4) channels of aggregation and luminescence. The Baseline and Gain are set with a single Pushbutton. The front panel has a 24 character Liquid Crystal Display (LCD) for each channel. The heater block temperature, the luminescence gain, the stirring speed and the operating mode (Optical or Impedance) as well as warning messages are displayed on the front panel LCD. The instrument has the ability to use Disposable or Reusable Electrodes for Impedance Aggregation.
Included with the Model 700 are software programs for platelet aggregation and the ristocetin cofactor assay. The AGGRO/LINK®8 program provides real time color display of up to four (4) channels of aggregation and ATP release, displaying a total of eight (8) traces. This program performs computation of the traces reporting the results as maximum amplitude and slope. Lag time and area under the curveanalysis are also available for each trace. Each graph, its values, patient demographics and reagent information are then stored on computer disk for later retrieval and printing. Test graphs can be easily recalled for viewing or printing, while data points can be copied to Excel.
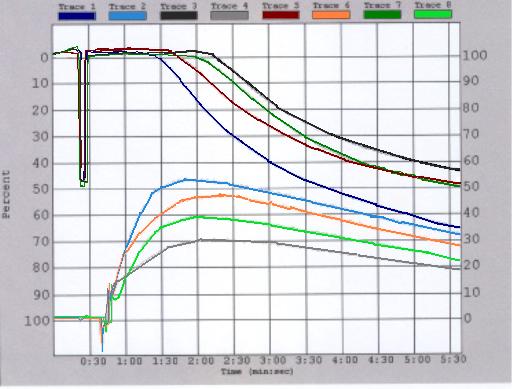
The CHRONO-LOG® von Willebrand Cofactor software package simplifies the vW assay. On-screen instructions guide the operator through the procedures to run dilutions for the standard curve and to test patient samples. The best-fit standard curve is calculated automatically and then stored along with reagent lot numbers. Up to four (4) samples can be processed simultaneously; tests on samples need not start and stop at the same time, allowing for stress-free operation. Percent activity is automatically calculated and reported.
The CHRONO-LOG® Model 700 instrument is FDA approved and CE marked and can be used for all clinical and pharmaceutical studies.